  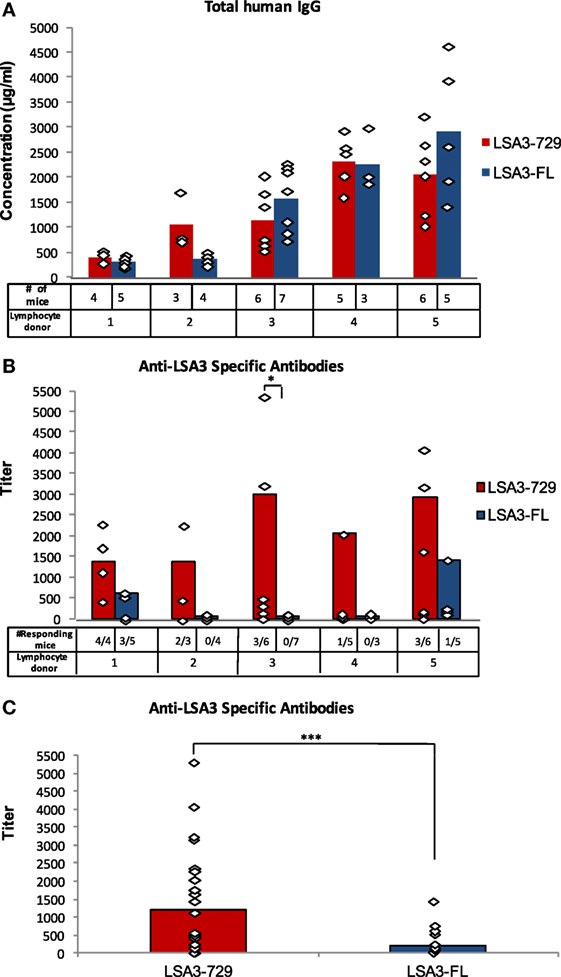
This lack of success has resulted in a bottleneck in the preclinical evaluation of drugs and in their therapeutic efficacy on primary human tumors in vivo, leaving a major knowledge gap on drug efficacy prior to clinical translation. To accomplish this, primary human tumors have been engrafted into numerous strains of immunodeficient mice, but until recently there has been varying and often poor success depending on the nature of the primary tumor and on the immunodeficient recipient used. Therefore, a longstanding goal for cancer investigators has been to engraft in an animal model primary patient-derived human tumors (referred to as patient-derived xenografts, or PDX) that maintain the genetic, phenotypic, and functional characteristics of the primary tumor after transplantation. In attempts to overcome these issues, human tumor cell lines and solid human tumors were first engrafted into nude mice and later into early models of scid mice, but these early models have shown limited success for investigating the pathogenesis of tumors and the efficacy of drugs on their growth and survival. This can be due to many factors, including genetic alterations that occur through multiple passages of the cell line, lack of appropriate human tumor stromal-associated cell populations that are important for tumor growth and maintenance, lack of an immunologic environment in which the tumor is exposed to in vivo, and issues associated with vascularization and the three-dimensional structure of the tumor in vivo as compared to a two-dimensional cell layer in vitro. However, many of the mechanisms responsible for tumorigenicity are altered during cell culture, and drugs that show efficacy in vitro against human tumor cell lines are often ineffective when translated to primary tumors in patients. The use of human tumor cell lines has led to increased understanding of the molecular events that lead to malignancy and fueled the promise of rapid advances in cancer therapy. Moreover, NSG mice can be engrafted with functional human immune systems permitting for the first time the potential to study primary human tumors in vivo in the presence of a human immune system. Programs to optimize patient-specific therapy using patient-derived xenograft (PDX) tumor growth in NSG mice have been established at several institutions, including The Jackson Laboratory. Using NSG mice as human xenograft recipients, it is now possible to grow almost all types of primary human tumors in vivo, including most solid tumors and hematological malignancies that maintain characteristics of the primary tumor in the patient. The IL2rγ null mutation has been used to develop several immunodeficient strains of mice, including the NOD- scid IL2rγ null (NSG) strain. More recently, mice deficient in adaptive immunity have been crossed with mice bearing targeted mutations designed to weaken the innate immune system, ultimately leading to the development of immunodeficient mice bearing a targeted mutation in the IL2 receptor common gamma chain gene ( IL2rγ null). These improvements include the discovery of the scid mutation and development of targeted mutations in the recombination activating genes 1 and 2 ( Rag1 null, Rag2 null) that severely cripple the adaptive immune response of the murine host. 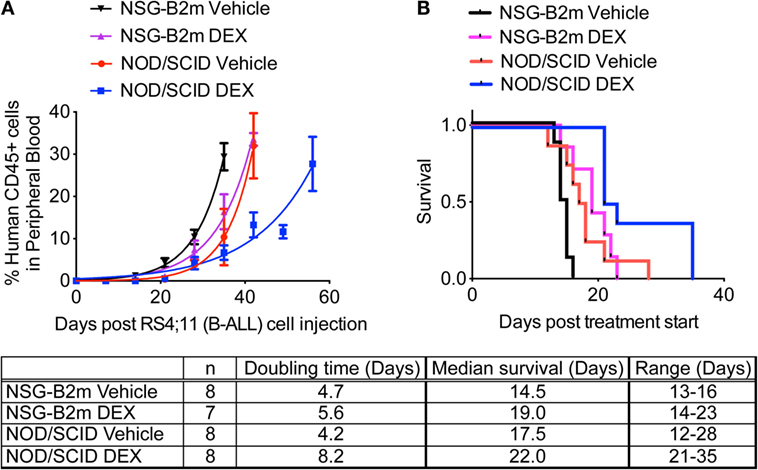
The field has advanced significantly over the ensuing years due to improvements in the murine recipient of human tumors. Since the discovery of the “nude” mouse over 40 years ago, investigators have attempted to model human tumor growth in immunodeficient mice. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
